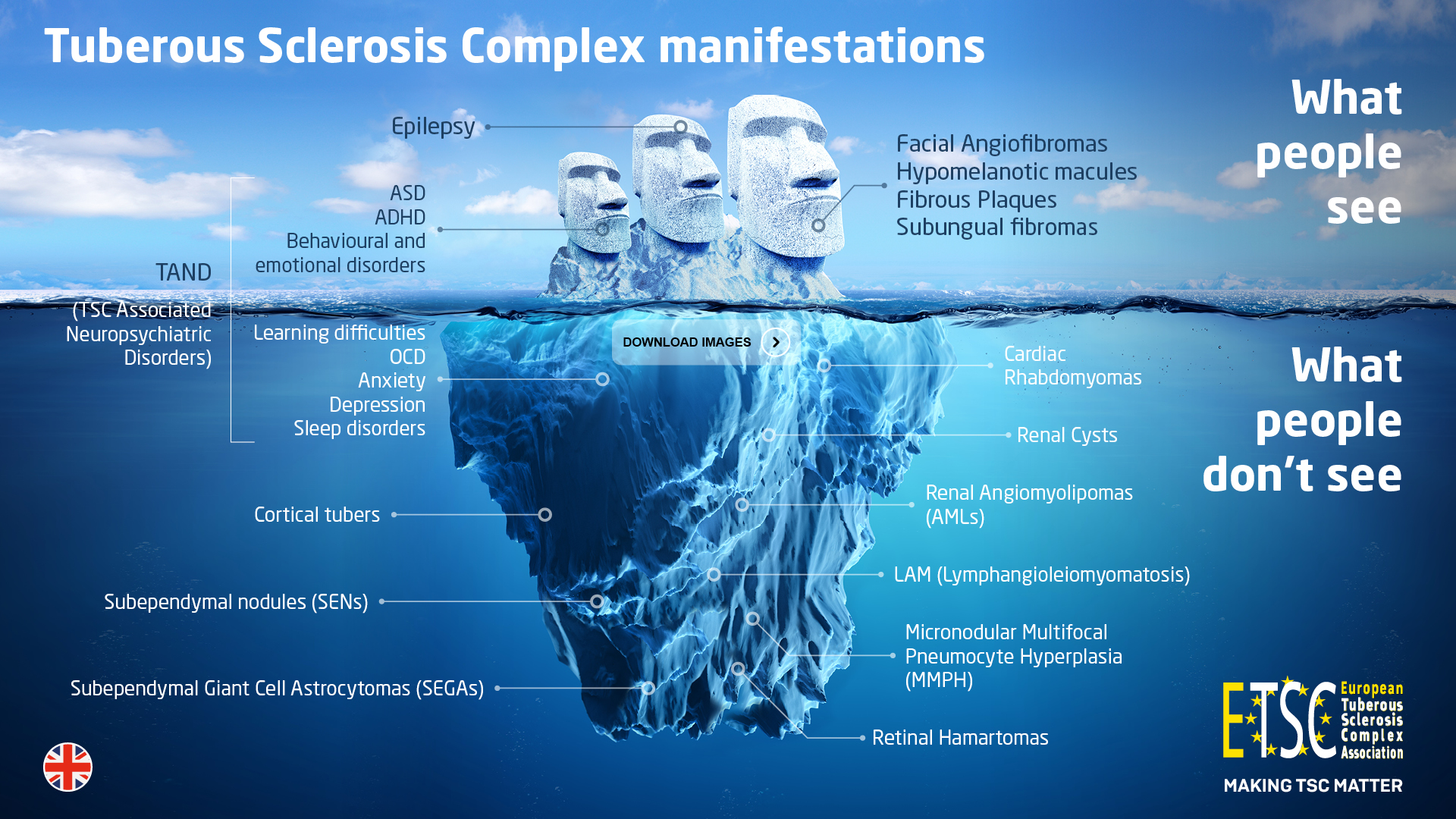
The Disease
Tuberous Sclerosis Complex (TSC) is a multi-system genetic disorder with a birth incidence of approximately 1 in every 6000.
It is estimated that one to two million people worldwide are affected.
TSC causes the growth of non-malignant tumours to form in vital organs.
TSC is also the leading genetic cause for epilepsy and autism.
TSC is:
Complex and variable
• Physical
– Tumours in the brain, heart, lungs, kidneys, skin and eyes
– Seizures
– Skin problems and facial angiofibromas
– Ungual fibromas, gum fibromas and dental pits
– TSC can also manifest itself in other organs
• In addition
– Cognitive impairment
– Behavioural issues
– Autism Spectrum Disorder (ASD)
– Sleep disorders
Needs support through whole life.
Requires many health and social experts to work together.
Needs specific centres of expertise dedicted to Tuberous Sclerosis Complex (TSC).
THERE IS NO CURE.

Useful Information
What is Tuberous Sclerosis Complex?
Tuberous Sclerosis Complex (TSC) is a rare, genetic condition causing the growth of benign tumours (non-cancerous) in various organs of the body, mainly the brain, kidneys, heart, eyes, lungs and skin. When it affects the central nervous system this can result in a combination of symptoms including seizures, developmental delay, cognitive impairment, autism spectrum disorder and behavioural problems; all these aspects strongly impact the quality of life of those affected including their carers.
The estimated incidence of the disorder is approximately one in 6,000 births.
Over one million people worldwide are estimated to have TSC.
TSC occurs in all races and ethnic groups, and in both genders.
The incidence and severity of TSC can vary widely between individuals—even between identical twins.
Individuals can be affected by different aspects of TSC at different times in their lives.
However, many people with TSC live independent, healthy lives and often go undiagnosed.
The last decade has seen much progress in the development of research in TSC so knowledge on the condition is now larger and increasing. Understanding does however vary from country to country.
Published documents and guidelines based on scientific evidence from key opinion leaders in TSC are now available.
What causes Tuberous Sclerosis Complex?
TSC is a genetic condition, meaning that it is caused by a change, or mutation, in a gene.
Two genes that cause TSC have been identified:TSC1 and TSC2.
The TSC1 gene is located on chromosome 9 and produces a protein called hamartin.
The TSC2 gene is located on chromosome 16 and produces the protein tuberin.
All genes including TSC1 and TSC2 come in pairs, a mutation in only one copy of these two genes can cause TSC. Therefore, TSC is a dominant condition because the change in one copy dominates over the other causing the condition and its symptoms.
When one copy of either TSC1 or TSC2 is mutated in a cell or group of cells, the other copy of that pair of genes becomes vulnerable to damage during normal cell division. If the other normal copy is mutated as well that cell loses control of its mTOR pathway and becomes a TSC cell.
In normal cells the mTOR pathway integrates messages from outside the cell including information on growth, division and how much nutrition is available to do so. When control is lost the TSC cell grows and divides in an uncontrolled manner and does not behave like a normal cell. i.e. it does not move to where it should be, become specialized to perform a particular function, nor die when it is no longer needed.
Diagnosis
TSC is usually diagnosed according to accepted clinical diagnostic criteria. Molecular genetic diagnosis of TSC by analysis of a blood sample is now available but these genes are very complex and their analysis can sometimes take a long time.
Approximately 80% of cases can be identified.
Where the family-specific mutation is identified, then early pre-natal diagnosis can be performed through a chorion villus biopsy.
Approximately 1 in 3 people with TSC have inherited a faulty gene from an affected parent via dominant inheritance. If the parent transmits a copy of the gene without the change, then the child will not have TSC. Therefore, there is a 50% chance with each pregnancy for a parent with TSC to have a child with TSC.
In 2 out of 3 children with TSC, neither parent is affected. In these cases it is thought the cause is a “sporadic” or new mutation, with this child being the first person in the family to have TSC. The cause of these new mutations is not known.
Mosaicism is a term used to describe a genetic alteration that does not occur in all body cells. The proportion of normal to abnormal cells will determine the severity of the disorder.
Sometimes, apparently physically unaffected parents of a child with TSC have a mutation only in either the ovaries or their testes; this is called germ-line mosaicism.
The chance of unaffected parents of a child with TSC having a second child with TSC is approximately 3%.
Organs affected
Brain involvement
Almost all individuals with TSC will have some signs of the condition in their brain. The most common neurological feature of TSC is epilepsy or seizures. Approximately 75% – 90% of individuals with TSC will have epilepsy at some point in their lives. Epilepsy can start at any age but many babies with TSC have a seizure during the first year of life.
TSC can cause three different types of brain lesions:
Cortical tubers
Subependymal nodules (SENs)
Subependymal giant cell astrocytomas (SEGAs )
Some people will have all the lesions, whereas others will have no brain involvement at all.
Cortical tubers
Cortical tubers are found on the surface of the brain and appear as an abnormal mass of tissue. These areas of the brain contain abnormal cells and are best visualised by Magnetic Resonance Imaging (MRI) of the brain. It is thought that tubers form during foetal development, so the number of tubers in the brain will stay the same throughout the life of the individual with TSC. They can sometimes be detected by MRI as early as 20 weeks gestation. Tubers can calcify or undergo cystic degeneration.
Subependymal nodules (SENs)
SENs are present in about 80% of people with TSC and are believed to be asymptomatic . SENS are nodular lesions that are usually less than 1cm in size. SENs develop along with the rest of the brain but they may calcify or become hard and can be easily detected with a computed tomography (CT) scan.
It is believed that some SENs can grow to form SEGAs, although the mechanism is still unknown.
Subependymal Giant Cell Astrocytomas (SEGAs)
Subependymal giant cell astrocytomas (SEGAs) are also called Subependymal giant cell tumors (SGCTs) and will develop in approximately 15% of individuals with TSC. They are located in the ventricles in the brain and usually appear in the first 20 years of life.
SEGAs are non-cancerous tumours, but if a giant cell astrocytoma grows large enough, it can block the flow of fluid inside the ventricles of the brain and the tumour will have to be removed and/or the ventricles shunted to relieve fluid build-up and pressure. This build-up of pressure can result in symptoms that include vomiting, nausea, and headaches as well as changes in appetite, behaviour, and mood, level of intellectual functioning or exacerbation of epilepsy. These symptoms may or may not signal tumour growth, but they can be interpreted as warnings that there may be a problem.
Brain imaging should be done at the time of diagnosis of TSC to obtain a baseline image and then at 1 to 3 year intervals.
A brain scan can sometimes show growth even before symptoms develop.
Intellectual ability and behavioural difficulties
Intellectual abilities of people affected with TSC can range from very high to very low.
Regular assessment of cognitive behaviour and development should be performed in order to evaluate any future changes.
If there is a change in cognitive development and behaviour then these changes should be assessed to identify and treat them. These changes could be due to exacerbation of seizures, sub-clinical seizures, new psychological problems such as anxiety and depression, new communication difficulties or an enlarging SEGA for example.
Behavioural issues including aggression, depression, sleep disorders, speech and language difficulties, repetitive behaviour etc. , are more common in people with TSC with an intellectual disability.
Heart involvement
Cardiac rhabdomyomas are non-cancerous tumours that occur in the heart in approximately 50% of babies born with TSC.
Cardiac rhabdomyomas may be detected during pregnancy, they are seen on prenatal foetus ultrasound exams but may not appear until the second half of pregnancy. They may be single or multiple and are often the first clinical sign that the baby has TSC. Cardiac rhabdomyomas are at their largest at the time of the baby's birth and generally get progressively smaller.
If they do not cause problems at birth they typically regress, or shrink during childhood and do not usually affect the individual in later life.
Occasionally, if the tumours are large or are multiple, they can block the circulation and in rare occasions lead to death. They may also cause dangerous arrhythmias. A few individuals will have long-term problems with heart rhythm that will need to be monitored throughout their lives.
The Kidney
It is estimated that approximately 80% of individuals with TSC will develop angiomyolipomas (AMLs) or cysts during their lifetime. Although most people with TSC will develop AMLs and won’t have major problems, some AMLs can effect the correct functioning of the kidney.
AMLs are benign growths consisting of fatty tissue, muscle cells and vascular cells . They begin to grow in childhood but may not cause a problem until early adulthood.
Once identified, it is important that AMLs are frequently monitored by a nephrologist.
AMLs can sometimes grow so large that they cause pain or kidney failure. Bleeding from AMLs may also occur causing both pain and weakness. Bleeding is rare in AMLs that are smaller than 3cm but the risk of bleeding increases as the size increases.
If bleeding does not stop naturally, there may be severe blood loss, resulting in profound anaemia and a life-threatening drop in blood pressure, warranting urgent medical attention.
Individuals with TSC may also have simple cysts in one or both kidneys, usually these cause no problems.
A small number (2%) of people with TSC also have polycystic kidneys disease (PKD), which can cause high blood pressure and impair kidney function.
The gene for PKD is right next to the TSC2 gene, severe PKD will usually be visible during childhood or even before birth .
Renal cell carcinoma is rare, estimated to be seen in 1-4% of individuals with TSC and will occur at a younger age than in the general population.
Oncocytomas are benign tumours only occasionally seen in individuals with TSC.
Other organs
Although cysts and AMLs occurring in other organs such as the pancreas, ovaries and liver are a rare occurrence, if problems should arise in these organs then treatment from the appropriate specialist should be sought.
The Skin
The skin signs of TSC are highly variable from one individual with TSC to the next, even within the same family.
Some skin signs appear at birth, others develop later in childhood or even adulthood.
Nearly all people with TSC will have at least one of the skin signs; for many people these are one of the first signs of TSC.
Some TSC skin signs are hardly noticeable, other individuals may have larger TSC skin signs that cause pain or bleed easily.
The different signs of TSC on the skin are:
- Hypomelanotic macules (ash-leaf spots)
Areas of skin that contain less pigment than surrounding skin and therefore they appear lighter in colour. Ultraviolet light is used to identify these as they may otherwise be hard to see in babies or people with fair skin. Almost all people with TSC have them; they may be present at birth and usually persist throughout life. They can be found anywhere on the skin.
If they are on the scalp, eyebrows or eyelashes they can cause a white patch of hair.
The hypomelanotic macules do not cause any symptoms or complications.
- Facial angiofibromas
These appear across the cheeks, nose and chin (sometimes they resemble acne), these small reddish bumps often appear at the age of 4 or 5 years and may increase in size with age. Facial angiofibromas are overgrowths of normal skin cells, composed of blood vessels (‘angio’) and fibrous tissue (‘fibroma’).
Laser therapy is recommended for improvement of facial angiofibromas. There are several types of lasers that can give good results; argon lasers, pulsed dye lasers and CO2 laser is recommended for the more fibrous type of facial angiofibromas.
Recent research has shown rapamycin to be effective in improving facial angiofibromas, but in some countries this can be difficult to obtain.
Research has also suggested that occurrence of new facial angiofibromas may be more likely if people are exposed to UV light, therefore applying 30 SPF cream sun to the skin should help prevent the rash getting worse.
- Shagreen Patches
These are usually found on the back or nape of the neck but may been seen on other parts of the body. This skin patch is an area of thickened and rough looking skin similar to orange peel.
It consists of an excess amount of fibrous tissue.
Shagreen patches can be treated with laser or plastic surgery.
- Fibrous Plaques
These are usually found on the forehead, but fibrous plaques may also occur on the cheeks or scalp.
Some individuals with TSC are born with these; some develop them gradually over the first 10 years of life. After that they generally stay the same size but they can become thicker over time.
- Nail Lesions
Ungual fibromas or subungual fibromas are smooth, firm growths that grow around or from underneath the fingernails or toenails.
These lesions may deform the nail by pushing it up from the nail bed causing infection and bleeding. Ungual fibromas on the toes can be painful when wearing shoes and may need to be surgically removed.
- Eyes
Nearly 50% of individuals with TSC have eye involvement (benign tumours).
These harmatomas are either single or multiple and most commonly located near to or at the disc margin of the retina, although they may be seen in the peripheral retina as well. Sometimes they are smooth-surfaced, these are called non-calcified harmatomas, others are shaped like a mulberry and are called calcified harmatomas. Sometimes they are in between and called transitional harmatomas. They very rarely change over time so usually no treatment is required.
Retinal hypo-pigmented lesions are lesions on the eyes that are similar to the white patches seen on the skin in many people with TSC.
Visual loss may be associated with retinal hamartomas, either due to retinal or optic nerve involvement. This may also occur from intracranial brain tumors that affect either the part of the brain that processes visual information, or increased pressure created in the brain and resulting in secondary injury to the optic nerve. Visual decline can also accompany subependymal giant cell astrocytomas associated with the optic nerve.
- Lungs
Signs of TSC in the lungs are mostly found in women and occur only after puberty. The lungs can be affected in two different ways with Lymphangioleiomyomatosis (LAM) and Micronodular Multifocal Pneumocyte Hyperplasia (MMPH).
Lymphangioleiomyomatosis (LAM) is a progressive lung complication of TSC caused by an overgrowth of cells in the airways, blood vessels and lymph vessels. The word lymphangioleiomyomatosis can be broken down into its parts to help explain what the disease is: lymph refers to the lymph vessels and angio refers to the blood vessels in the body, while leiomyomatosis refers to the formation of the unusual TS cells in the lung, which look like a smooth muscle (called LAM cells). When LAM cells spread through the lung they destroy the lungs and make it difficult for oxygen to get across the wall of the airways and into the blood cells. It can cause chest pain, spontaneous pneumothorax or insidious dyspnea .
LAM occurs almost exclusively in women and in TSC. Some studies estimated that LAM will occur in 30-40% of women with TSC.
As it occurs mainly in women it is thought that it is oestrogen related.
Micronodular Multifocal Pneumocyte Hyperplasia (MMPH) can be found in women and men with TSC and usually does not affect respiratory function, it causes the overgrowth (hyperplasia) of the pneumocytes (a specific type of cell found in the lining of the air sacs in the lung) into small nodules.
MMPH is a benign condition but it is sometimes mistaken for other lung metastasis (spread of cancer).
A high resolution CT scan is recommended to all women with TSC at the age of approximately 18.
- Teeth and mouth
People with TSC may have dental pits and an overgrowth of the gums (fibromas) meaning teeth and gum tissue are slightly different and will require a little more care to keep them healthy. The tooth enamel will have random pits that occur on their surface, decay may be triggered here. A dentist can easily see and repair the enamel pits.
Good dental hygiene, including regular brushing and flossing is important.
How is TSC treated?
There is no cure for TSC although treatment is available for a number of the symptoms.
Anti-epileptic drugs may be used to control seizures. Vigabatrin is a particularly useful medication in TSC and has been approved by the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA).
Research into the investigation of mTOR inhibitors for the reversal of the underlying defects in TS cells is ongoing.
Respiratory insufficiency due to LAM can be treated with supplemental oxygen therapy or lung transplantation if severe.
The potential use of mTOR inhibitors for other problems (epilepsy, autism spectrum disorder, neuro-cognition and facial angiofibromas (topically) is still under investigation.
Specific medications may be prescribed for behavioural issues. Intervention programs including assistance at school and occupational therapy may benefit individuals with special needs and developmental issues.
Surgery may be needed where complications connected to tubers, SENs or SEGAs, as well as for the risk of haemorrhage from kidney tumours are concerned.
Because TSC is a lifelong condition, individuals need to be regularly monitored by TSC specialists preferably in a TSC clinic to ensure they are receiving the best possible treatments. Due to the many varied symptoms of TSC, care by a clinician with experience in the disorder is recommended.
What is the prognosis?
The prognosis for individuals with TSC is highly variable and depends on the severity of symptoms. Those individuals with mild symptoms usually do well and have a normal life expectancy, while paying attention to TSC-specific issues. Individuals who are severely affected can suffer from severe intellectual impairment and persistent (refractory) epilepsy.
All individuals with TSC are at risk for life-threatening conditions related to the brain tumours, kidney lesions, or LAM.
Continued monitoring by a physician with experience of TSC is important.
With appropriate medical care, most individuals with the disorder can look forward to a normal life expectancy.
2012 International TSC Clinical Consensus Conference Recommendations
http://tscinternational.org/documents/TSCi%20Consensus%20Guidelines.pdf
RECOMMENDATIONS FOR NEWLY DIAGNOSED INDIVIDUALS
Published diagnostic criteria provide healthcare professionals with guidelines for the diagnosis of TSC since each clinical feature is not TSC specific.
Recommendations for newly diagnosed individuals of any age
- Review the newly diagnosed individual’s nearest three generations (siblings, parents, and either children or grandparents). Genetic testing for family counselling or when TSC diagnosis is in question should be offered.
- Undergo magnetic resonance imaging (MRI) of the brain to look for possible subependymal giant cell astrocytomas (SEGAs), subependymal nodules (SENs), and tubers.
- Have an assessment for TSC-associated neuropsychiatric disorder (TAND), a new terminology to describe the interrelated behavioural, intellectual, and neuropsychiatric features common in TSC.
- Obtain a baseline routine electroencephalogram (EEG); if EEG is abnormal, and particularly if features of TAND are present, follow this with 24-hour video EEG to look for subtle seizure activity.
- Perform MRI of the abdomen to check for possible renal angiomyolipomas or cysts. Kidney function (glomerular filtration rate, or GFR) and blood pressure should be measured.
- Undergo dermatological and dental examinations to check for abnormalities of the skin and teeth that are frequently associated with TSC.
- Perform a routine electrocardiogram (ECG) to check for abnormal heart rhythm.
- Perform an echocardiogram to assess cardiac function and presence of rhabdomyomas (especially in children under 3 years of age).
- Undergo an exam by an ophthalmologist for possible vision problems or abnormalities of the retina.
- Additional recommendations for newly diagnosed infants and children under 3 years old
- Teach parents and other caregivers of children under 3 years of age about how to recognize infantile spasms and what to do if they suspect the child is having infantile spasms.
Additional recommendations for newly diagnosed adults (18 years of age or older)
- Perform baseline pulmonary function testing and high-resolution computed tomography (HRCT) in adult females 18 years of age or older to check for possible lymphangioleiomyomatosis (LAM). Younger females and adult males should only be evaluated for LAM when clinical symptoms are present that heighten suspicion (such as unexplained chronic cough, chest pain, or breathing difficulties).
RECOMMENDATIONS FOR INDIVIDUALS ALREADY DIAGNOSED WITH TSC
Recommendations for individuals of any age
- Offer genetic testing (if not done previously) and family counseling to affected individuals upon reaching reproductive age.
- Obtain EEG in individuals with known or suspected seizures. The duration and frequency of EEG should be determined by clinical need rather than set or defined ages or intervals.
- Treat seizures other than infantile spasms similarly to that for other types of epilepsy. In individuals with TSC whose seizures are resistant to commonly used anti-seizure medications, the ketogenic/low- glycemic diet, vagus nerve stimulation, and epilepsy surgery can be of benefit.
- Screen for TAND symptoms at each clinical visit. Any findings of concern should prompt more detailed evaluation and treatment. In addition, formal behavioural, intellectual, and neuropsychiatric evaluation should be performed at least once during each key developmental stage: 0-3 years old, 3- 6 years old, 6-9 years old, 12-16 years old, and 18-25 years old. TAND symptoms should be treated with a combination strategy of pharmacologic and non-pharmacologic interventions, individualized for the specific TAND profile of each patient.
For the TAND checklist, please click here: TAND Checklist
- Perform MRI of the brain every 1-3 years until age 25 years even in asymptomatic individuals to monitor for emergence or progression of SEGA. The frequency of MRI should be increased if SEGA is large or growing. Adults with SEGA in childhood may continue to require periodic MRIs. When a SEGA is causing symptoms of fluid accumulation in the brain, surgical removal of the SEGA is the preferred treatment when possible. Growing SEGA that are not causing symptoms can be treated with surgery or mTOR inhibitors.
- Obtain abdominal MRI every 1-3 years to monitor renal and non-renal TSC disease progression.
- Check blood pressure and glomerular filtration rate at least annually.
- Treat angiomyolipomas associated with acute bleeding by vascular embolization and corticosteroids.
- Angiomyolipomas without acute bleeding that are larger than 3 cm in diameter and growing should be treated with an mTOR inhibitor as first-line therapy to prevent continued growth and bleeding; embolisation and corticosteroids or kidney-sparing resection are appropriate second-line therapies.
- Examine skin annually for new or worsening TSC-associated lesions. Severe or problematic lesions may be treated by surgery, laser, or topical mTOR inhibitors.
- Perform a dental examination twice per year by a dentist experienced with recognition and management of dental issues common in TSC.
- Undergo a detailed eye and vision examination, which is repeated if clinically indicated in individuals with previously identified retinal lesions or new vision complaints or concerns. Individuals treated with vigabatrin should also undergo periodic ophthalmologic evaluations.
- Obtain an echocardiogram every 1-3 years in individuals with previously identified cardiac rhabdomyomas until regression/stabilization of cardiac rhabdomyomas is established.
- Obtain an ECG every 3-5 years to check for problems with electrical activity in the heart. Recommendations for infants and children under 3 years old.
- Treat infantile spasms with vigabatrin as first-line therapy. Adrenocorticotropic hormone (ACTH) can be used as second-line therapy if vigabatrin treatment is unsuccessful.
Recommendations for adults (18 years of age or older)
- Perform clinical screening for LAM symptoms, including exertional dyspnea and shortness of breath, at each clinical visit.
- Obtain HRCT every 5-10 years in asymptomatic persons at risk for LAM (i.e. all adult females 18 years of age or older and males or females of any age with clinical symptoms present that heighten suspicion). Patients with previously identified LAM should obtain HRCT more frequently (every 2-3 years) to monitor for disease progression.
- Obtain pulmonary function testing annually in patients with previously identified LAM or if new respiratory difficulties or concerns arise in previously asymptomatic persons at risk for LAM.
Disclaimer
We have made every effort to ensure that the information in this website is correct and up-to-date at the time of publication.
However, research is ongoing and clinical practice may differ slightly from country to country. Therefore, ETSC cannot accept responsibility for any errors.
About Us
Established in 2012, ETSC is a federation of Tuberous Sclerosis Associations.
ETSC believes in total “patient experience” and that interaction with all healthcare providers and relative agencies is paramount.
Objectives
• Increase knowledge and awareness on TSC
• Promote implementation of European and International diagnostic criteria, surveillance and treatment guidelines
• Stimulate research on TSC
• Interest European and International organisations in the welfare of those with TSC and their families
• Exchange information of mutual interest between associations.
• Total patient experience and interaction in each individual country with all healthcare providers and agencies
• Uniting TSC Associations to ensure knowledge and surveillance on our rare disorder and orphan drugs are increased
• Good working relationships with Pharma and the inclusion of representatives from National Organisations in information and education on Clinical trials and any new drugs being developed for use with our rare disease
• Vigilance on protocols and new drugs
E-TSC e.V European Tuberous Sclerosis Complex Association
Walkmuehlstr. 1 – 65195 Wiesbaden – Germany
Registered NPO Nr. VR6685 Registry Court of Wiesbaden
Board of Directors: Carla Fladrowski (President), Micaela Rozenberg President & Treasurer), Helmut Hehn (General Secretary), Tom Carter, Inna Korob, Adelheid Wiemer-Kruel; Chris Kingswood (Honory Member)
Bank account for donations: IBAN DE23 6605 0101 0108 1974 35, BIC KARSDE66XXX, Sparkasse Karlsruhe

DEA
DEA
Disseminating Expertise and Achievement across the EU
Aim
To create a readily accessible, active and sustainable network within and between EU countries, for people affected by Tuberous Sclerosis Complex.
Project Outline
E-TSC has determined that across Europe there is a need to identify Key Opinion Leaders (KOL) in each individual country treating patients affected by Tuberous Sclerosis Complex (TSC). A network is also required which will facilitate the sharing of best practice within a relevant clinical speciality as well as across disciplines when the need arises in this multi-system condition.
The project aims to establish a holistic approach to the treatment of TSC and promote multi-disciplinary working across the EU.
At present (largely informal) professional practice demonstrates:
- networking at national and international scientific conferences in each specific discipline
- networking at PO meetings
- professional networks
ETSC considers the following main issues:
- considerable variation between countries
- considerable variation between individual practitioners – KOL may individually be high profile, but grass roots practitioners may not have contact with any other professional with experience of TSC
- (perceived) lack of succession - need to build up TSC expertise in newly qualified professionals.
The project is intended as a catalyst to promote and facilitate future communication between specialists in each particular area related to TSC locally and across the EU, encouraging shared best practice, timely awareness of innovative therapies and dissemination and adherence to International treatment guidelines. In this way the well-being and quality of life of people with TSC will be enhanced.
Methodology
"Pit Stops" (expertise sharing sessions) shall be organised in parallel to local TSC Associations' Annual General Meetings. Each Pit Stop will be hosted by a known KOL identified by the hosting association plus other identified experts and interns in the specific field.
In these face to face meetings specialists will each be invited to present a case study that they wish to discuss with colleagues. 1 or 2 expert patients should be present at each Pit Stop.
ETSC will also be attending specialised Congresses across Europe ( European Nephrology; Neurology, Psychiatry, Dermatology etc.) and recruiting the interest of specialists in that particular field.
An important component of all Pit Stops is the opportunity given to patients to speak to the experts.
DEA Pit Stops
Athens, Greece - September 2014
Porto, Portugal - September 2014
Madrid, Spain - March 2015
Milan, Italy - October 2014
Lisbon, Portugal - April 2015
Bari, Italy - October 2015
Kehl-Kork, Germany - April 2016
Moscow, Russia - November 2016
Budapest, Hungary - October 2017
Leuven, Belgium - October 2018
Project Leaders:
Carla Fladrowski
Micaela Rozenberg
To read a message from the Board of Directors os ETSC, please click here.
TAND
Prof Petrus J de Vries
Sue Struengmann Professor of Child & Adolescent Psychiatry
Division of Child & Adolescent Psychiatry
Department of Psychiatry and Mental Health
University of Cape Town, South Africa
Prof Anna Jansen
Head of Clinics, Paediatric Neurology Unit, Department of Paediatrics,
UZ Brussels, Belgium
Members

Austria


Belgium


Denmark


Finland

France


Germany


Greece


Ireland


Israel


Italy


Macedonia

Netherlands


Norway


Poland


Portugal


Russia


Serbia


Slovakia


Spain

Sweden


Switzerland


Ukraine


United Kingdom


Hungary

Events & News
Contact Us
Walkmuehlstr. 1
65195 Wiesbaden,
GERMANY